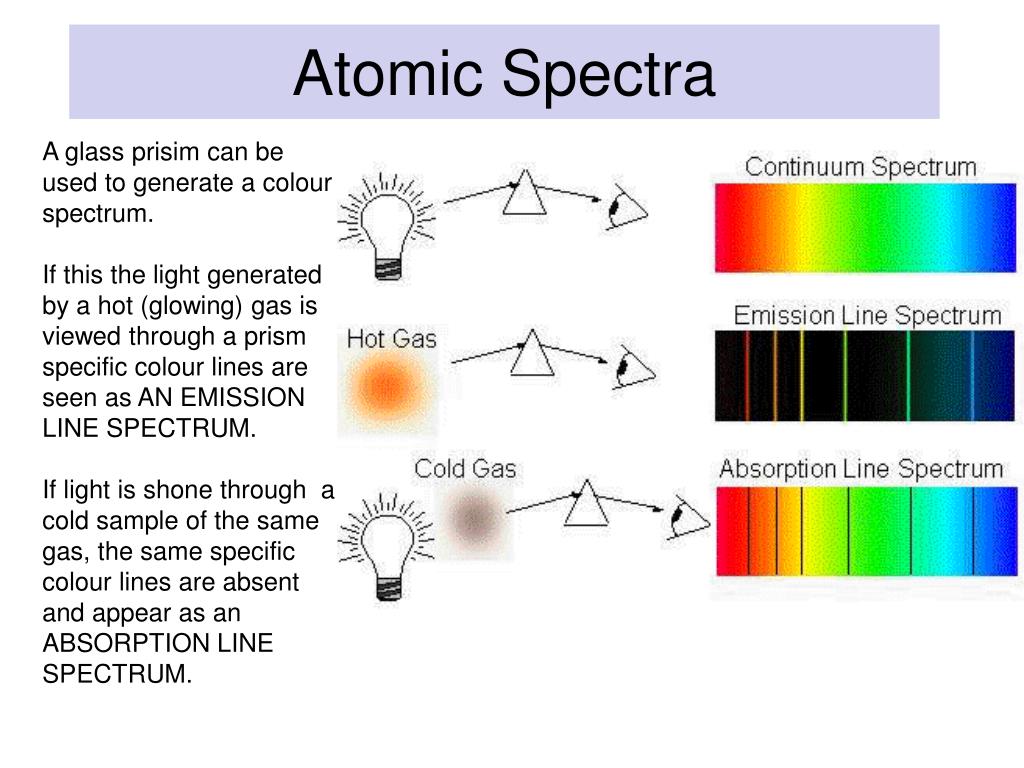
This describes the effect on a beam of white light passing through a cool (unstimulated) sample of vapour of a particular element. The emission spectrum shown has 4 distinct bright spectral lines or colours with frequencies corresponding to quantum electron jumps from one of four excited states down to the ground state. In the case of the hydrogen atoms, in the example above each individual atom in the sample has five potential quantised energy bands, only one of which is occupied by an electron at any time. 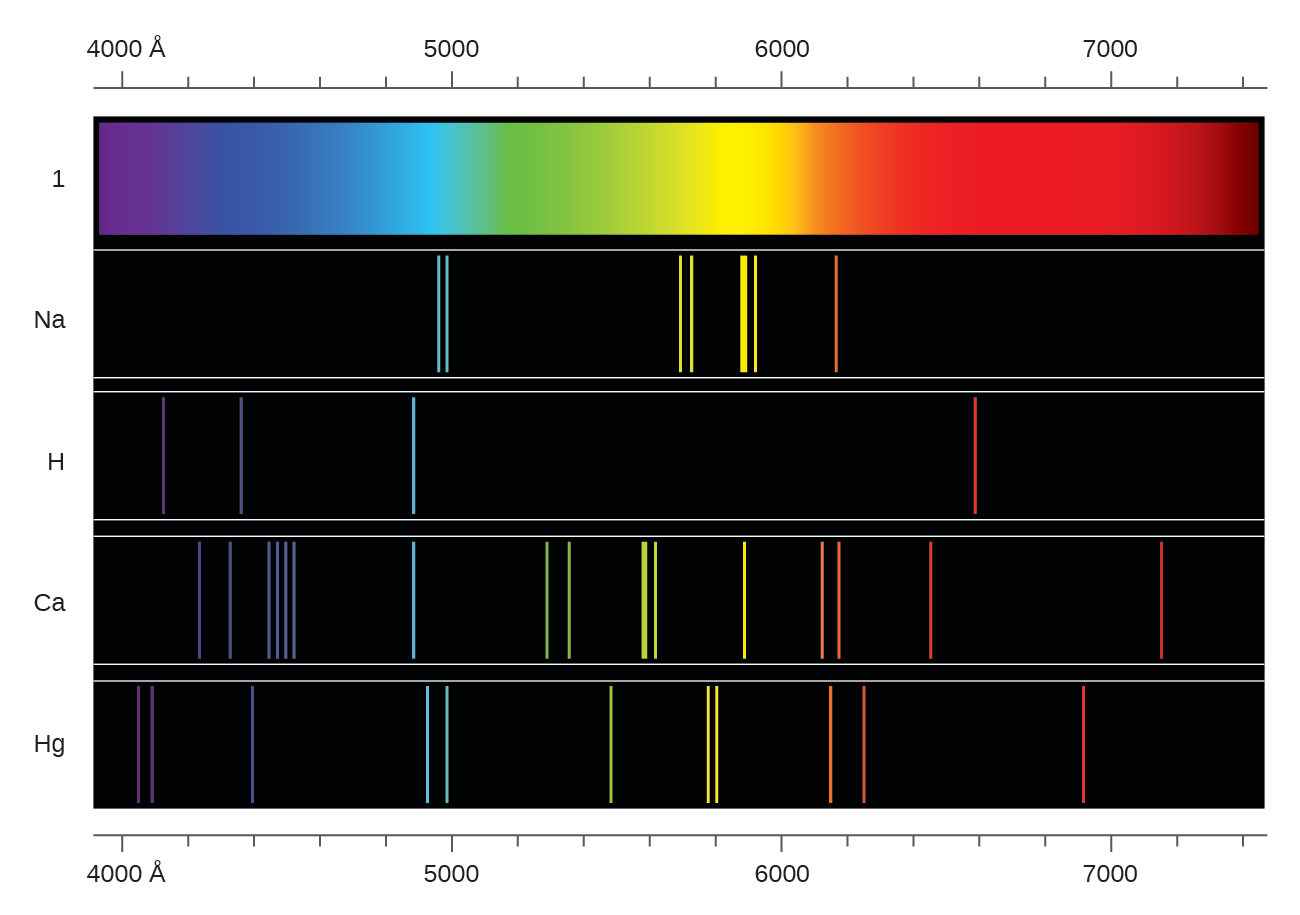
This is the light generated by stimulating a source consisting of a sample of single element type. White light consists of a continuous spectrum of frequencies resulting from so called black body radiation which encompasses radiation emitted from multiple different elements and the electron jumps between their multiple energy bands. In the hydrogen spectrum opposite, the spectral line with a wavelength of 656 nm (or frequency of 457,000 GHz) corresponds to an energy difference of 4.5 x 10 -5 eV between the electron's ground state and its excited state, known as the fine structure enrgy gap. They are the frequencies associated with the energy released or absorbed when an electron jumps from one discrete energy level to another. The spectral lines of light emitted by an atom are known as its characteristic frequencies. The dark lines in the Sun's radiation spectrum were first catalogued by Fraunhofer in 1814 and subsequently investigated by Kirchhoff and Bunsen who provided the initial analysis an explanation of atomic spectral lines. The corresponding wavelength λ is given by λ = c/f where c is the speed of light. The frequency f of the light energy released or absorbed in the quantum jump is given by Planck's Law E= hf where E is the difference in energy levels between the initial orbit and the final orbit and h is Planck's constant. Heat or other stimuli may cause an electron to break out of its energy band and jump over one or more energy bands down to a lower energy orbit releasing a photon with a quantum of energy corresponding to the difference in energy level between the two orbits.Ĭonversely, when photons collide with an atom, their energy may be transferred to and absorbed by the atom's electrons causing them to jump to a higher energy level orbit. N is also known as the quantum number representing the energy state of the atom. progressively further away from the nucleus have correspondingly higher energy levels. The shell or orbit n1 closest to the nucleus is known as the ground state and has the lowest energy level. The shells and the electrons in them have characteristic quantised energy levels. Each shell may carry one or more electrons or none at all. Woodbank does not monitor or record these emailsīohr's model of the atom envisaged a nucleus around which electrons circulate in one or more distinct orbits (also called shells or energy bands or more recently orbitals).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
